How to reduce the risk of atherosclerosis?Target cholesterol
Atherosclerosis is characterized by lipid accumulation, inflammatory response, cell death and fibrosis in the arterial wall, which is the pathological basis for ischemic coronary heart disease (CHD) and stroke. Major risk factors for atherosclerosis include high plasma levels of low-density lipoprotein (LDL) cholesterol and low plasma concentrations of high-density lipoprotein (HDL) cholesterol. Intervention studies with statins have shown to reduce plasma LDL cholesterol concentrations and subsequently the risk of developing CHD. However, not all the aggressive statin therapy could decrease the risk of developing CHD. Therefore, additional therapeutic interventions beyond statins are strongly needed to further reduce the risk of developing CHD and stroke. Recently, multiple studies have reported the research on the improvement of AS by targeting cholesterol, which may provide help for the prevention and treatment of cardiovascular diseases.
1. Epsin Nanotherapy Regulates Cholesterol Transport to Fortify Atheroma Regression
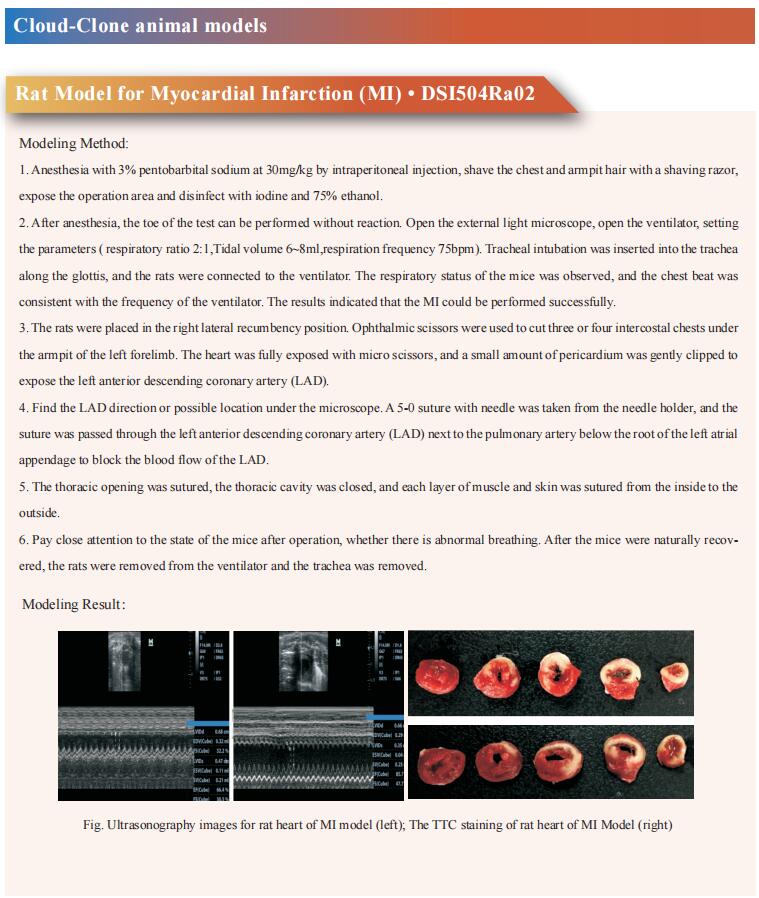
Excess cholesterol accumulation in lesional macrophages elicits complex responses in atherosclerosis. Epsins, a family of endocytic adaptors, fuel the progression of atherosclerosis; however, the underlying mechanism and therapeutic potential of targeting Epsins remains unknown. Hong Chen, Vascular Biology Program, Boston Children's Hospital and Department of Surgery, Harvard Medical School, USA, and his team revealed that Epsins regulate lipid metabolism and transport in atherosclerotic macrophages[1]. Inhibiting Epsins by nanotherapy halts inflammation and accelerates atheroma resolution. Harnessing lesional macrophage-specific nanoparticle delivery of Epsin small-interfering RNAs, they showed that silencing of macrophage Epsins diminished atherosclerotic plaque size and promoted plaque regression. Mechanistically, they demonstrated that Epsins bound to CD36 to facilitate lipid uptake by enhancing CD36 endocytosis and recycling. Conversely, Epsins promoted ABCG1 degradation via lysosomes and hampered ABCG1-mediated cholesterol efflux and reverse cholesterol transport (Fig.1). These findings suggest that targeting Epsins in lesional macrophages may offer therapeutic benefits for advanced atherosclerosis by reducing CD36-mediated lipid uptake and increasing ABCG1-mediated cholesterol efflux.
2. Intracellular tPA-PAI-1 interaction determines VLDL assembly in hepatocytes
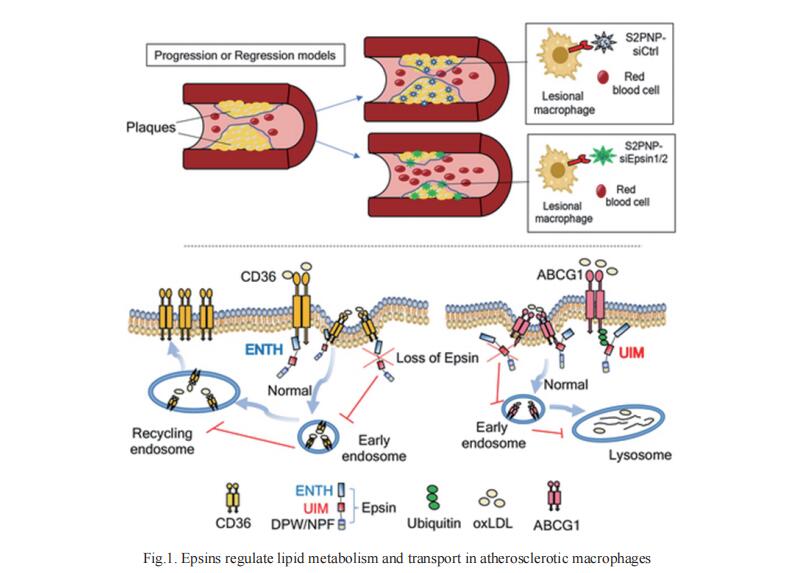
Apolipoprotein B (apoB)–lipoproteins initiate and promote atherosclerotic cardiovascular disease.Plasma tissue plasminogen activator (tPA) activity is negatively associated with atherogenic apoBlipoprotein cholesterol levels in humans, but the mechanisms are unknown. Ze Zheng, Department of Medicine, Medical College of Wisconsin, USA, and his team found that tPA,partially through the lysine-binding site on its Kringle 2 domain, binds to the N terminus of apoB,blocking the interaction between apoB and microsomal triglyceride transfer protein (MTP) in hepatocytes,thereby reducing very-low-density lipoprotein (VLDL) assembly and plasma apoB-lipoprotein cholesterollevels (Fig.2)[2]. Plasminogen activator inhibitor 1 (PAI-1) sequesters tPA away from apoB and increases VLDLassembly. Humans with PAI-1 deficiency have smaller VLDL particles and lower plasma levels of apoBlipoprotein cholesterol. This study reveals that the tPA-PAI-1 interaction determines VLDL assembly in hepatocytes. These findings provide mechanisticinsight into the physiologic response to lipidingestion and to the atherosclerosis-relevantprocess of excessive apoB-lipoprotein production by the liver.
3. Smooth muscle α-actin missense variant promotes atherosclerosis through modulation of intracellular cholesterol in smooth muscle cells
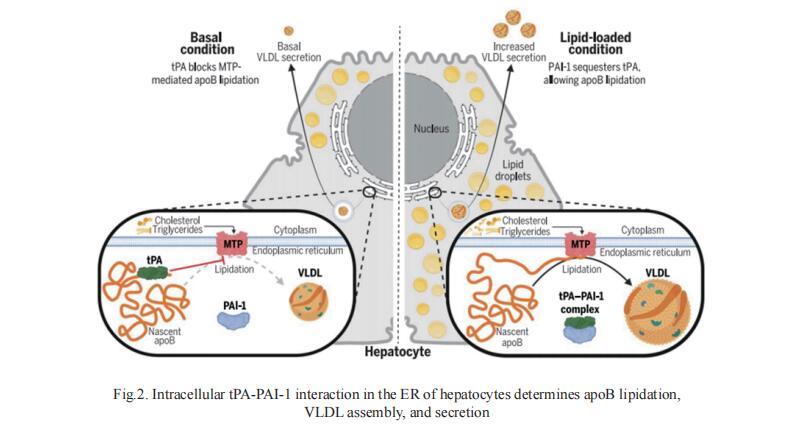
The variant p.Arg149Cys in ACTA2, which encodes smooth muscle cell (SMC)-specific α-actin, predisposes to thoracic aortic disease and early onset coronary artery disease in individuals without cardiovascular risk factors. Dianna M Milewicz, Division of Medical Genetics, Department of Internal Medicine, McGovern Medical School, The University of Texas Health Science Center at Houston, USA, and his team investigated how this variant drives increased atherosclerosis[3]. Hyperlipidemic Acta2R149C/+Apoe−/− mice have a 2.5-fold increase in atherosclerotic plaque burden compared to Apoe−/− mice with no differences in serum lipid levels. At the cellular level, misfolding of the R149C α-actin activates heat shock factor 1, which increases endogenous cholesterol biosynthesis and intracellular cholesterol levels through increased HMG-CoA reductase (HMG-CoAR) expression and activity. The increased cellular cholesterol in Acta2R149C/+ SMCs induces endoplasmic reticulum stress and activates PERK-ATF4-KLF4 signaling to drive atherosclerosis-associated phenotypic modulation in the absence of exogenous cholesterol, while WT cells require higher levels of exogenous cholesterol to drive phenotypic modulation. These data identify a novel mechanism by which a mutant SMC contractile protein, SMA, predisposes to early onset CAD in individuals without hypercholesterolemia. And further highlight the role of cholesterol-induced phenotypic modulation of SMCs in driving atherosclerotic plaque formation.
References
[1]Cui K, Gao X, Wang B, et al. Epsin Nanotherapy Regulates Cholesterol Transport to Fortify Atheroma Regression. Circ Res. 2023;132(1):e22-e42. (IF=20.1)
[2]Dai W, Zhang H, Lund H, et al. Intracellular tPA-PAI-1 interaction determines VLDL assembly in hepatocytes. Science. 2023;381(6661):eadh5207. (IF=56.9)
[3]Kaw K, Chattopadhyay A, Guan P, et al. Smooth muscle α-actin missense variant promotes atherosclerosis through modulation of intracellular cholesterol in smooth muscle cells. Eur Heart J. 2023;44(29):2713-2726. (IF=39.3)
Cloud-Clone can not only provide multiple cardiovascular system disease models, including hyperlipidemia and atherosclerosis, hypertension, myocardial hypertrophy, myocardial infarction, arrhythmia, heart failure, etc., covering common cardiovascular system diseases. It also has all kinds of common indicators of cardiovascular system signal pathway and inflammatory indicators related products, which can help the majority of scientific researchers to study cardiovascular system diseases.