Packages (Simulation)

Reagent Preparation
Image (I)
Image (II)
Certificate


ELISA Kit for Angiotensin I Converting Enzyme 2 (ACE2)
ACEH; ACEII; ACE-II; Peptidyl-Dipeptidase A; ACE-related carboxypeptidase; Angiotensin-converting enzyme homolog; Metalloprotease MPROT15
- Product No.SEB886Ra
- Organism SpeciesRattus norvegicus (Rat) Same name, Different species.
- Sample TypeSerum, plasma, tissue homogenates and other biological fluids
- Test MethodDouble-antibody Sandwich
- Assay Length3h
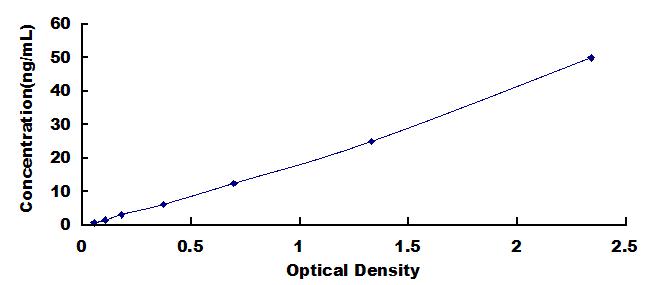
- Detection Range0.78-50ng/mL
- SensitivityThe minimum detectable dose of this kit is typically less than 0.31ng/mL.
- DownloadInstruction Manual
- UOM 48T96T 96T*5 96T*10 96T*100
- FOB
US$ 505
US$ 722
US$ 3249
US$ 6137
US$ 50540
For more details, please contact local distributors!
Specificity
This assay has high sensitivity and excellent specificity for detection of Angiotensin I Converting Enzyme 2 (ACE2).
No significant cross-reactivity or interference between Angiotensin I Converting Enzyme 2 (ACE2) and analogues was observed.
Recovery
Matrices listed below were spiked with certain level of recombinant Angiotensin I Converting Enzyme 2 (ACE2) and the recovery rates were calculated by comparing the measured value to the expected amount of Angiotensin I Converting Enzyme 2 (ACE2) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 99-105 | 102 |
| EDTA plasma(n=5) | 79-93 | 80 |
| heparin plasma(n=5) | 78-95 | 86 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Angiotensin I Converting Enzyme 2 (ACE2) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Angiotensin I Converting Enzyme 2 (ACE2) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Angiotensin I Converting Enzyme 2 (ACE2) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 84-92% | 83-97% | 80-95% | 97-104% |
| EDTA plasma(n=5) | 80-102% | 91-101% | 78-104% | 83-105% |
| heparin plasma(n=5) | 88-104% | 85-92% | 98-105% | 78-101% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| Pre-coated, ready to use 96-well strip plate | 1 | Plate sealer for 96 wells | 4 |
| Standard | 2 | Standard Diluent | 1×20mL |
| Detection Reagent A | 1×120µL | Assay Diluent A | 1×12mL |
| Detection Reagent B | 1×120µL | Assay Diluent B | 1×12mL |
| TMB Substrate | 1×9mL | Stop Solution | 1×6mL |
| Wash Buffer (30 × concentrate) | 1×20mL | Instruction manual | 1 |
Assay procedure summary
1. Prepare all reagents, samples and standards;
2. Add 100µL standard or sample to each well. Incubate 1 hours at 37°C;
3. Aspirate and add 100µL prepared Detection Reagent A. Incubate 1 hour at 37°C;
4. Aspirate and wash 3 times;
5. Add 100µL prepared Detection Reagent B. Incubate 30 minutes at 37°C;
6. Aspirate and wash 5 times;
7. Add 90µL Substrate Solution. Incubate 10-20 minutes at 37°C;
8. Add 50µL Stop Solution. Read at 450nm immediately.
GIVEAWAYS
INCREMENT SERVICES
-
 Single-component Reagents of Assay Kit
Single-component Reagents of Assay Kit
-
 Lysis Buffer Specific for ELISA / CLIA
Lysis Buffer Specific for ELISA / CLIA
-
 Quality Control of Kit
Quality Control of Kit
-
 ELISA Kit Customized Service
ELISA Kit Customized Service
-
 Disease Model Customized Service
Disease Model Customized Service
-
 Serums Customized Service
Serums Customized Service
-
 TGFB1 Activation Reagent
TGFB1 Activation Reagent
-
 Real Time PCR Experimental Service
Real Time PCR Experimental Service
-
 Streptavidin
Streptavidin
-
 Fast blue Protein Stain solution
Fast blue Protein Stain solution
-
 Single-component Reagents of FLIA Kit
Single-component Reagents of FLIA Kit
-
 Streptavidin-Agarose Beads
Streptavidin-Agarose Beads
| Magazine | Citations |
| Nature Medicine | Angiotensin-Converting Enzyme 2 Overexpression Remarkably Ameliorated Glomerular Injury in a Rat Model of Diabetic Nephropathy: A Comparison with ACE Inhibition MolMed: 10_11_liu |
| Journal of Cancer Therapy | Plasma Levels of Angiotensin-Converting Enzymes 1 and 2 and AGTR2 (T1247G and A5235G) Gene Polymorphisms Are Associated to Breast Cancer Progression. Scirp: Source |
| Reprod Toxicol.? | ACE2 activation by xanthenone prevents leptin-induced increases in blood pressure and proteinuria during pregnancy in Sprague-Dawley rats Pubmed:25205467 |
| Intensive Care Medicine Experimental | Angiotensin converting enzymes in patients with acute respiratory distress syndrome Content: 3 |
| Journal of Chromatography A | Application of a new procedure for liquid chromatography/mass spectrometry profiling of plasma amino acid-related metabolites and untargeted shotgun proteomics to identify mechanisms and biomarkers of calcific aortic stenosis S0021967317311858 |
| Journal of Clinical Pharmacy and Therapeutics | Relationship between genetic variants of ACE2 gene and circulating levels of ACE2 and its metabolites. pubmed:28895159 |
| Innate Immunity | Lipoxin A attenuates LPS-induced acute lung injury via activation of the ACE2-Ang-(1-7)-Mas axis Pubmed:29969931 |
| Journal of Surgical Research | Impact of angiotensin-converting enzyme 2 levels on postoperative pneumonia after esophagectomy Pubmed:29506841 |
| canadian journal of physiology and pharmacology | Could cardioprotective effect of ACE2 activator “diminazene aceturate” is more potent than ACE inhibitor “Enalapril” on acute myocardial infarction in rats? Pubmed: 30840489 |
| Intensive Care Medicine | Angiotensin-converting enzymes in acute respiratory distress syndrome |
| BioMed Research International | AVE 0991 Attenuates Pyroptosis and Liver Damage after Heatstroke by Inhibiting the ROS-NLRP3 Inflammatory Signalling Pathway |
| Heart and vessels | Relationship between circulating levels of angiotensin-converting enzyme 2-angiotensin-(1–7)-MAS axis and coronary heart disease Pubmed: 31359146 |
| Medicine | Alteration and association between serum ACE2/angiotensin (1-7)/Mas axis and oxidative stress in chronic kidney disease: A pilot study Pubmed: 32756181 |
| American Journal of Respiratory and Critical Care Medicine | Equilibrium Angiotensin Metabolite Profiling in Patients with Acute Respiratory Distress Syndrome Indicates Angiotensin Converting Enzyme Inhibition Pubmed: 32628511 |
| Clinical Research in Cardiology | Determinants of soluble angiotensin-converting enzyme 2 concentrations in adult patients with complex congenital heart disease 33280062 |
| Clinical & Translational Immunology | Pattern of circulating SARS©\CoV©\2©\specific antibody©\secreting and memory B©\cell generation in patients with acute COVID©\19 33552508 |
| researchsquare | Novel transgenic mice with Cre-dependent co-expression of GFP and human ACE2: a safe tool for study of COVID-19 pathogenesis 33855640 |
| Viruses | Different Neutralization Sensitivity of SARS-CoV-2 Cell-to-Cell and Cell-Free Modes of Infection to Convalescent Sera |
| Sci Rep | Immunoreactivity of the SARS-CoV-2 entry proteins ACE-2 and TMPRSS-2 in murine models of hormonal manipulation, ageing, and cardiac injury 34907257 |
| European Journal of Clinical Investigation | Angiotensin‐converting enzyme 2 and Transmembrane protease serine 2 in female and male patients with end‐stage kidney disease Pubmed:35366343 |
| Metabolites | Ramipril Reduces Acylcarnitines and Distinctly Increases Angiotensin-Converting Enzyme 2 Expression in Lungs of Rats Pubmed:35448480 |