Packages (Simulation)

Reagent Preparation
Image (I)
Image (II)
Certificate


High Sensitive ELISA Kit for Prothrombin Fragment 1+2 (F1+2)
- Product No.HEA710Hu
- Organism SpeciesHomo sapiens (Human) Same name, Different species.
- Sample TypeSerum, plasma and other biological fluids
- Test MethodDouble-antibody Sandwich
- Assay Length3h
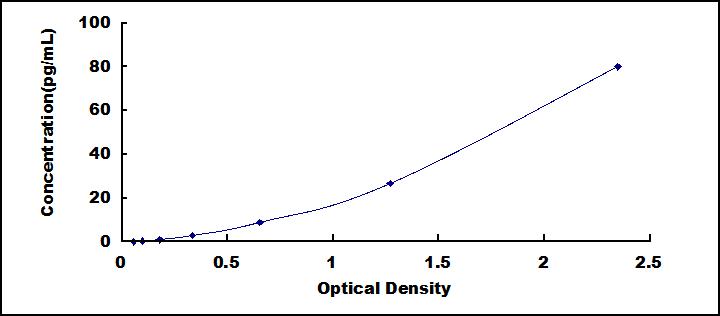
- Detection Range0.11-80pg/mL
- SensitivityThe minimum detectable dose of this kit is typically less than 0.04pg/mL.
- DownloadInstruction Manual
- UOM 48T96T 96T*5 96T*10 96T*100
- FOB
US$ 490
US$ 700
US$ 3150
US$ 5950
US$ 49000
For more details, please contact local distributors!
Specificity
This assay has high sensitivity and excellent specificity for detection of High Sensitive Prothrombin Fragment 1+2 (F1+2).
No significant cross-reactivity or interference between High Sensitive Prothrombin Fragment 1+2 (F1+2) and analogues was observed.
Recovery
Matrices listed below were spiked with certain level of recombinant High Sensitive Prothrombin Fragment 1+2 (F1+2) and the recovery rates were calculated by comparing the measured value to the expected amount of High Sensitive Prothrombin Fragment 1+2 (F1+2) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 81-99 | 95 |
| EDTA plasma(n=5) | 90-101 | 93 |
| heparin plasma(n=5) | 89-96 | 93 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level High Sensitive Prothrombin Fragment 1+2 (F1+2) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level High Sensitive Prothrombin Fragment 1+2 (F1+2) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of High Sensitive Prothrombin Fragment 1+2 (F1+2) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 87-103% | 91-105% | 83-90% | 93-105% |
| EDTA plasma(n=5) | 91-101% | 85-99% | 82-89% | 87-94% |
| heparin plasma(n=5) | 97-105% | 79-98% | 90-104% | 78-101% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| Pre-coated, ready to use 96-well strip plate | 1 | Plate sealer for 96 wells | 4 |
| Standard | 2 | Standard Diluent | 1×20mL |
| Detection Reagent A | 1×120µL | Assay Diluent A | 1×12mL |
| Detection Reagent B | 1×120µL | Assay Diluent B | 1×12mL |
| TMB Substrate | 1×9mL | Stop Solution | 1×6mL |
| Wash Buffer (30 × concentrate) | 1×20mL | Instruction manual | 1 |
Assay procedure summary
1. Prepare all reagents, samples and standards;
2. Add 100µL standard or sample to each well. Incubate 1 hours at 37°C;
3. Aspirate and add 100µL prepared Detection Reagent A. Incubate 1 hour at 37°C;
4. Aspirate and wash 3 times;
5. Add 100µL prepared Detection Reagent B. Incubate 30 minutes at 37°C;
6. Aspirate and wash 5 times;
7. Add 90µL Substrate Solution. Incubate 10-20 minutes at 37°C;
8. Add 50µL Stop Solution. Read at 450nm immediately.
GIVEAWAYS
INCREMENT SERVICES
| Magazine | Citations |
| Critical Care | Disseminated intravascular coagulation or acute coagulopathy of trauma shock early after trauma? An observational study BioMed: cc10553 |
| Journal of Vascular Surgery | Changes in thrombin generation, fibrinolysis, platelet and endothelial cell activity, and inflammation following endovascular abdominal aortic aneurysm repair ScienceDirect: S0741521411018568 |
| Scandinavian Journal of Trauma, Resuscitation and Emergency Medicine | High levels of soluble VEGF receptor 1 early after trauma are associated with shock, sympathoadrenal activation, glycocalyx degradation and inflammation in severely injured patients: a prospective study Sjtrem:1757-7241 |
| Journal of Thrombosis and Haemostasis | High sCD40L levels early after trauma are associated with enhanced shock, sympathoadrenal activation, tissue and endothelial damage, coagulopathy and mortality Wiley: source |
| British Journal of Dermatology | Evaluation of autologous plasma skin test in patients with chronic idiopathic urticaria PubMed: 21910697 |
| Arteriosclerosis, Thrombosis, and Vascular Biology. | Diet Modulates Endogenous Thrombin Generation, A Biological Estimate of Thrombosis Risk, Independently of the Metabolic Status PubMed: 22859493 |
| Journal of Vascular Surgery | Medium-term effect of endovascular and open abdominal aortic aneurysm repair on thrombin generation and fibrinolysis PubMed: 23140799 |
| PLoS ONE | Markers of Thrombogenesis and Fibrinolysis and Their Relation to Inflammation and Endothelial Activation in Patients with Idiopathic Pulmonary Arterial Hypertension Plosone: Source |
| Journal of Vascular Surgery | Effect of endovascular and open abdominal aortic aneurysm repair on thrombin generation and fibrinolysis Pubmed: 23140799 |
| Haemetology | Clinical impact of factor V Leiden, prothrombin G20210A, and MTHFR C677T mutations among sickle cell disease patients of Central India Pubmed: 23992124 |
| Cogent Medicine | Study the association of adiponectin with inflammation and hypercoagulability in case of type 2 diabetic subjects with renal dysfunction Tandfonline:Source |
| Interact Cardiovasc Thorac Surg. | An ex vivo evaluation of blood coagulation and thromboresistance of two extracorporeal circuit coatings with reduced and full heparin dose Pubmed:24632424 |
| Perfusion | Ex vivo simulation of cardiopulmonary bypass with human blood for hemocompatibility testing PubMed: 26243277 |
| J Thromb Haemost | Comparison of the Effect of Dabigatran and Dalteparin on Thrombus Stability in a Murine Model of Venous Thromboembolism PubMed: 26514101 |
| Acta Oncologica Turcica | Thrombin activatable fibrinolysis inhibitor (TAFI), tissue factor pathway inhibitor (TFPI), and prothrombin fragment 1+2 levels in patients with advanced colorectal cancer aot:AOT_49_1_6_12.pdf |
| Advances in Medical Sciences | Perioperative thrombocytopenia predicts poor outcome in patients undergoing transcatheter aortic valve implantation pubmed:29145170 |
| Thrombosis Research | The causes of thrombocytopenia after transcatheter aortic valve implantation pubmed:28582640 |
| Acta Haematologica | Elevations of Thrombotic Biomarkers in Hemoglobin H Disease Pubmed:29402840 |
| Journal of Neurotrauma | The cerebral thrombin system is activated after intracerebral hemorrhage and contributes to secondary lesion growth and poor neurological outcome in C57Bl/6 mice Pubmed: 31830857 |
| Indian Journal of Public Health Research & Development | Enhanced Fibrin-Lysis in Grade-1 Dengue Haemorrhagic Fever |
| Rheumatol Int | The impact of anti-endothelial cell antibodies (AECAs) on the development of blood vessel damage in patients with systemic lupus erythematosus: the preliminary … Pubmed:35284968 |