Packages (Simulation)

Reagent Preparation
Image (I)
Image (II)
Certificate


- Featured-Product
ELISA Kit for Interleukin 1 Receptor Antagonist (IL1RA)
IL1RN; IL1-RA; ICIL1RA; IL1ra3; IL1F3; IRAP;IL-1RA; Interleukin-1 Family Member 3; IL1 inhibitor; Anakinra
- Product No.SEA223Mu
- Organism SpeciesMus musculus (Mouse) Same name, Different species.
- Sample Typeserum, plasma, tissue homogenates and other biological fluids
- Test MethodDouble-antibody Sandwich
- Assay Length3h
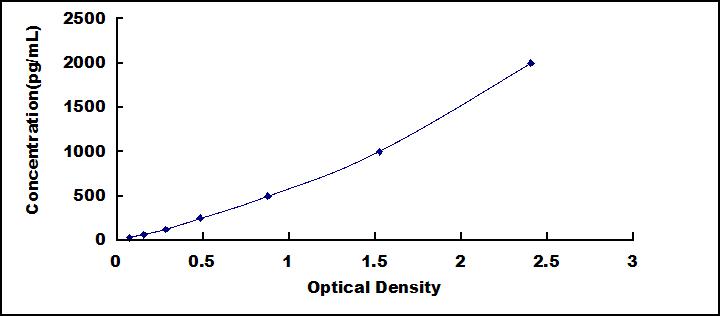
- Detection Range31.2-2,000pg/mL
- SensitivityThe minimum detectable dose of this kit is typically less than 11.5pg/mL.
- DownloadInstruction Manual
- UOM 48T96T 96T*5 96T*10 96T*100
- FOB
US$ 328
US$ 468
US$ 2106
US$ 3978
US$ 32760
For more details, please contact local distributors!
Specificity
This assay has high sensitivity and excellent specificity for detection of Interleukin 1 Receptor Antagonist (IL1RA).
No significant cross-reactivity or interference between Interleukin 1 Receptor Antagonist (IL1RA) and analogues was observed.
Recovery
Matrices listed below were spiked with certain level of recombinant Interleukin 1 Receptor Antagonist (IL1RA) and the recovery rates were calculated by comparing the measured value to the expected amount of Interleukin 1 Receptor Antagonist (IL1RA) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 79-90 | 83 |
| EDTA plasma(n=5) | 88-96 | 92 |
| heparin plasma(n=5) | 82-95 | 89 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Interleukin 1 Receptor Antagonist (IL1RA) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Interleukin 1 Receptor Antagonist (IL1RA) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Interleukin 1 Receptor Antagonist (IL1RA) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 94-102% | 89-101% | 97-105% | 81-90% |
| EDTA plasma(n=5) | 97-105% | 83-97% | 80-96% | 94-105% |
| heparin plasma(n=5) | 94-102% | 97-104% | 80-99% | 78-94% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| Pre-coated, ready to use 96-well strip plate | 1 | Plate sealer for 96 wells | 4 |
| Standard | 2 | Standard Diluent | 1×20mL |
| Detection Reagent A | 1×120µL | Assay Diluent A | 1×12mL |
| Detection Reagent B | 1×120µL | Assay Diluent B | 1×12mL |
| TMB Substrate | 1×9mL | Stop Solution | 1×6mL |
| Wash Buffer (30 × concentrate) | 1×20mL | Instruction manual | 1 |
Assay procedure summary
1. Prepare all reagents, samples and standards;
2. Add 100µL standard or sample to each well. Incubate 1 hours at 37°C;
3. Aspirate and add 100µL prepared Detection Reagent A. Incubate 1 hour at 37°C;
4. Aspirate and wash 3 times;
5. Add 100µL prepared Detection Reagent B. Incubate 30 minutes at 37°C;
6. Aspirate and wash 5 times;
7. Add 90µL Substrate Solution. Incubate 10-20 minutes at 37°C;
8. Add 50µL Stop Solution. Read at 450nm immediately.
GIVEAWAYS
INCREMENT SERVICES
-
 Single-component Reagents of Assay Kit
Single-component Reagents of Assay Kit
-
 Lysis Buffer Specific for ELISA / CLIA
Lysis Buffer Specific for ELISA / CLIA
-
 Quality Control of Kit
Quality Control of Kit
-
 ELISA Kit Customized Service
ELISA Kit Customized Service
-
 Disease Model Customized Service
Disease Model Customized Service
-
 Serums Customized Service
Serums Customized Service
-
 TGFB1 Activation Reagent
TGFB1 Activation Reagent
-
 Real Time PCR Experimental Service
Real Time PCR Experimental Service
-
 Streptavidin
Streptavidin
-
 Fast blue Protein Stain solution
Fast blue Protein Stain solution -
 Single-component Reagents of FLIA Kit
Single-component Reagents of FLIA Kit
-
 Streptavidin-Agarose Beads
Streptavidin-Agarose Beads
| Magazine | Citations |
| The Veterinary Journal | Mucosal imbalance of interleukin-1β and interleukin-1 receptor antagonist in canine inflammatory bowel disease ScienceDirect: S1090023312000962 |
| Daily quercetin supplementation over a period of two weeks results in a moderate accumulation of total plasma flavonols in horses Researchgate:Source | |
| Journal of Equine Veterinary Science | Synovial Fluid and Serum Concentrations of Interleukin-1 Receptor Antagonist and Interleukin-1 Science: Article |
| Journal of Equine Veterinary Science | A Two-Week Quercetin Supplementation in Horses Results in Moderate Accumulation of Plasma Flavonol Concentrations Science: Article |
| Scientific publications in peer reviewed journals | Evidence-based review of efficacy and adverse effects of joint medication and evaluation of synovial fluid and serum markers for osteoarthritis in the horse receive:101658 |
| Multiple Sclerosis and Related Disorders | Interleukin 1 receptor antagonist and 2′-5′-oligoadenylate synthetase-like molecules as novel biomarkers for multiple sclerosis patients in Bahrain pubmed:29141788 |
| Advances in Medical Sciences | A similar pro/anti-inflammatory cytokine balance is present in the airways of competitive athletes and non-exercising asthmatics pubmed:28822267 |
| Archives of Medical Research | Winter ambient training conditions are associated with increased bronchial hyperreactivity and with shifts in serum innate immunity proteins in young … Pubmed:29379533 |
| Journal of Veterinary Internal Medicine | Effect of interleukin‐1β on occludin mRNA expression in the duodenal and colonic mucosa of dogs with inflammatory bowel disease Pubmed:29572935 |
| Frontiers in Pharmacology | M10, a Myricetin-3-ObD-Lactose Sodium Salt, Prevents Ulcerative Colitis Through Inhibiting Necroptosis in Mice Pubmed: 33041798 |