Packages (Simulation)

Reagent Preparation
Image (I)
Image (II)
Certificate


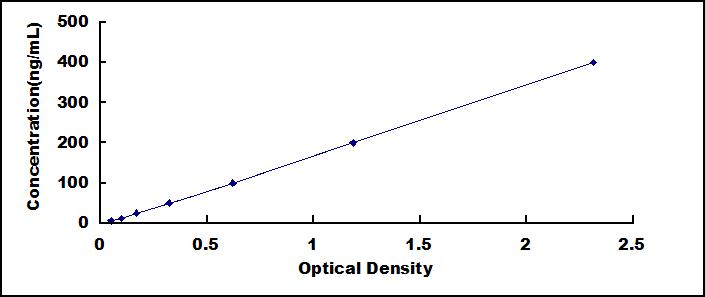
ELISA Kit for Serum Amyloid A (SAA)
SA-A; SAA1; PIG4; TP53I4; Amyloid protein A; Amyloid fibril protein AA; Serum amyloid A-1 protein
- Product No.SEA885Bo
- Organism SpeciesBos taurus; Bovine (Cattle) Same name, Different species.
- Sample Typeserum, plasma and other biological fluids
- Test MethodDouble-antibody Sandwich
- Assay Length3h
- Detection Range6.25-400ng/mL
- SensitivityThe minimum detectable dose of this kit is typically less than 2.72ng/mL.
- DownloadInstruction Manual
- UOM 48T96T 96T*5 96T*10 96T*100
- FOB
US$ 529
US$ 756
US$ 3402
US$ 6426
US$ 52920
For more details, please contact local distributors!
Specificity
This assay has high sensitivity and excellent specificity for detection of Serum Amyloid A (SAA).
No significant cross-reactivity or interference between Serum Amyloid A (SAA) and analogues was observed.
Recovery
Matrices listed below were spiked with certain level of recombinant Serum Amyloid A (SAA) and the recovery rates were calculated by comparing the measured value to the expected amount of Serum Amyloid A (SAA) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 88-101 | 96 |
| EDTA plasma(n=5) | 86-105 | 92 |
| heparin plasma(n=5) | 86-97 | 93 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Serum Amyloid A (SAA) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Serum Amyloid A (SAA) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Serum Amyloid A (SAA) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 82-96% | 81-102% | 81-103% | 91-102% |
| EDTA plasma(n=5) | 90-98% | 81-90% | 98-105% | 88-104% |
| heparin plasma(n=5) | 78-92% | 81-97% | 85-99% | 79-97% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| Pre-coated, ready to use 96-well strip plate | 1 | Plate sealer for 96 wells | 4 |
| Standard | 2 | Standard Diluent | 1×20mL |
| Detection Reagent A | 1×120µL | Assay Diluent A | 1×12mL |
| Detection Reagent B | 1×120µL | Assay Diluent B | 1×12mL |
| TMB Substrate | 1×9mL | Stop Solution | 1×6mL |
| Wash Buffer (30 × concentrate) | 1×20mL | Instruction manual | 1 |
Assay procedure summary
1. Prepare all reagents, samples and standards;
2. Add 100µL standard or sample to each well. Incubate 1 hours at 37°C;
3. Aspirate and add 100µL prepared Detection Reagent A. Incubate 1 hour at 37°C;
4. Aspirate and wash 3 times;
5. Add 100µL prepared Detection Reagent B. Incubate 30 minutes at 37°C;
6. Aspirate and wash 5 times;
7. Add 90µL Substrate Solution. Incubate 10-20 minutes at 37°C;
8. Add 50µL Stop Solution. Read at 450nm immediately.
GIVEAWAYS
INCREMENT SERVICES
-
 Single-component Reagents of Assay Kit
Single-component Reagents of Assay Kit
-
 Lysis Buffer Specific for ELISA / CLIA
Lysis Buffer Specific for ELISA / CLIA
-
 Quality Control of Kit
Quality Control of Kit
-
 ELISA Kit Customized Service
ELISA Kit Customized Service
-
 Disease Model Customized Service
Disease Model Customized Service
-
 Serums Customized Service
Serums Customized Service
-
 TGFB1 Activation Reagent
TGFB1 Activation Reagent
-
 Real Time PCR Experimental Service
Real Time PCR Experimental Service
-
 Streptavidin
Streptavidin
-
 Fast blue Protein Stain solution
Fast blue Protein Stain solution
-
 Single-component Reagents of FLIA Kit
Single-component Reagents of FLIA Kit
-
 Streptavidin-Agarose Beads
Streptavidin-Agarose Beads
| Magazine | Citations |
| Nitric Oxide. | Effect of feeding a high fat diet on hydrogen sulfide (H 2 S) metabolism in the mouse Pubmed:24637018 |
| Open Biochem J. | Acute Phase Response: Implication in ST-segment Elevation Myocardial Infarction Pubmed:24894970 |
| Iran J Pharm Res. | Comparison of the Effects of Enoxaparin and Heparin on Inflammatory Biomarkers in Patients with ST-segment Elevated Myocardial Infarction: A prospective Open Label Pilot Clinical Trial Pubmed:25237354 |
| US National Library of Medicine National Institutes of Health | No association between vitamin D levels and inflammation markers in patients with acute coronary syndrome Pubmed:25615956 |
| BMC Complementary and Alternative Medicine | Identification of plasma protein markers common to patients with malignant tumour and Abnormal Savda in Uighur medicine: a prospective clinical study Pubmed:25652121 |
| BOSCA | EFFECT OF UTERINE IMMUNOMODULATION ON SERUM AMYLOID-A CONCENTRATION AND CONCEPTION RATE IN CYCLIC NON BREEDING COWS Tcrjournals:Source |
| Life Sciences | Impaired adipogenesis in adipose tissue associated with hepatic lipid deposition induced by chronic inflammation in mice with chew diet PubMed: 26188592 |
| Endocrine | Relation of thyroid hormone abnormalities with subclinical inflammatory activity in patients with type 1 and type 2 diabetes mellitus PubMed: 26049370 |
| J Assist Reprod Genet. | The effect of serum and follicular fluid amyloid-associated protein levels on in vitro fertilization outcome in patients with polycystic ovary syndrome PubMed: 26463878 |
| International Journal of Clinical and Experimental Medicine | Circulating serum amyloid A: a potential biomarker for infection after aneurysmal subarachnoid hemorrhage files:ijcem0021201 |
| PLoS One. | An Anti-β-Amyloid Vaccine for Treating Cognitive Deficits in a Mouse Model of Down Syndrome Pubmed:27023444 |
| United States Patent Application | PATHWAY SPECIFIC MARKERS FOR DIAGNOSING IRRITABLE BOWEL SYNDROME y2016:0139148.html |
| Veterinary World | Acute phase response in lame crossbred dairy cattle pubmed:27956769 |
| Nature Communications | A ligand-specific blockade of the integrin Mac-1 selectively targets pathologic inflammation while maintaining protective host-defense Pubmed:29410422 |
| Journal of Animal Sciences | Influence of yeast culture and feed antibiotics on ruminal fermentation and site and extent of digestion in beef heifers fed high grain rations Pubmed:30060086 |
| Journal of Animal Science | Using ruminally protected and nonprotected active dried yeast as alternatives to antibiotics in finishing beef steers: growth performance, carcass traits, blood … Pubmed: 30184125 |
| Journal of Dairy Science | Metabolism and immune status during transition period influences the lactation performance in Zebu (Bos indicus () cows Pubmed: 17430930 |
| European Review for Medical and Pharmacological Sciences | The predictive value of joint detection of serum amyloid protein A, PCT, and Hs-CRP in the diagnosis and efficacy of neonatal septicemia. Pubmed: 31298341 |
| Research in Veterinary Science | Evaluation of infrared thermography compared to rectal temperature to identify illness in early postpartum dairy cows |
| Journal of Animal Science | Ruminally protected and unprotected Saccharomyces cerevisiae fermentation products as alternatives to antibiotics in finishing beef steers Pubmed: 31410465 |
| PLoS One | Effects of vaccination on acute-phase protein response in broiler chicken Pubmed: 32191767 |
| VETERINARY PATHOLOGY | AA Amyloid Deposition in the Central and Peripheral Nervous Systems in Flamingos Pubmed: 32677873 |
| Comp Immunol Microbiol Infect Dis | Serum amyloid A as an marker of cow֨ s mastitis caused by Streptococcus sp. Pubmed: 32505957 |
| Indian Journal of Animal Sciences | Peripheral concentrations of metabolic and inflammatory indicators during transition period and their relationship with postpartum clinical endometritis in dairy … |
| РОЛЬ СЫВОРОТОЧНОГО АМИЛОИДА А И С-РЕАКТИВНОГО БЕЛКА В ПРОГНОЗИРОВАНИИ ПОСЛЕОПЕРАЦИОННЫХ ОСЛОЖНЕНИЙ И … | |
| JOURNAL OF ANIMAL SCIENCE | Processing Index of Barley Grain and Dietary Undigested Neutral Detergent Fiber Concentration Affected Chewing Behavior, Ruminal pH and Total Tract Nutrient?¡ 33474564 |
| Animals | Induction of Serum Amyloid A3 in Mouse Mammary Epithelial Cells Stimulated with Lipopolysaccharide and Lipoteichoic Acid 34070499 |
| peerj | Perfluorooctane sulfonate exerts inflammatory bowel disease-like intestinal injury in rats 33510972 |
| Arquivo Brasileiro de Medicina Veterin¨¢ria e Zootecnia | CCS e amiloide A do leite de quartos mam¨¢rios tratados na secagem com plasma rico em plaquetas aut¨®logo, associado ou n?o a antibi¨®tico |
| Probiotics Antimicrob Proteins | The Effect of Feed Supplementation with EM Bokashi® Multimicrobial Probiotic Preparation on Selected Parameters of Sow Colostrum and Milk as Indicators … 34596883 |
| Vet Anim Sci | Effects of dietary turmeric (Curcuma longa) on innate and acquired immune responses in broiler chicken 34765801 |
| Brain Sci | Mahanimbine Improved Aging-Related Memory Deficits in Mice through Enhanced Cholinergic Transmission and Suppressed Oxidative Stress, Amyloid Levels, and … Pubmed:35053756 |
| J Clin Med | A Three-Protein Panel to Support the Diagnosis of Sepsis in Children Pubmed:35329889 |
| Diabetes Metabolic Syndrome and Obesity-Targets and Therapy | Serum and salivary adipokines in type 2 diabetes–Results of a pilot study in India Pubmed:35717896 |
| Clinical and Experimental Immunology | Clinical significance of differential serum-signatures for early prediction of severe dengue among Eastern Indian patients Pubmed:35348620 |
| İneklerde Subklinik Mastitis Tanısında Akut Faz Proteinlerinden Amiloid A ve C-Reaktif Protein Düzeylerinin Değerlendirilmesi |
| Catalog No. | Related products for research use of Bos taurus; Bovine (Cattle) Organism species | Applications (RESEARCH USE ONLY!) |
| RPA885Bo01 | Recombinant Serum Amyloid A (SAA) | Positive Control; Immunogen; SDS-PAGE; WB. |
| PAA885Bo01 | Polyclonal Antibody to Serum Amyloid A (SAA) | WB |
| MAA885Bo21 | Monoclonal Antibody to Serum Amyloid A (SAA) | WB; IHC; ICC; IP. |
| SEA885Bo | ELISA Kit for Serum Amyloid A (SAA) | Enzyme-linked immunosorbent assay for Antigen Detection. |
| LMA885Bo | Multiplex Assay Kit for Serum Amyloid A (SAA) ,etc. by FLIA (Flow Luminescence Immunoassay) | FLIA Kit for Antigen Detection. |